Demonstrating the Power of TWO2 Therapy
Level 1/A Randomized Controlled Trials
+
Real-World Outcomes
=
strong, Durable Wound Healing Like None Other
Chronic lower-extremity wounds persist because of a self-reinforcing cycle of inflammation, edema, and tissue hypoxia, a cycle that Standard of Care alone often can’t resolve. TWO₂ therapy (intermittent Topical Wound Oxygen therapy) was designed to break this cycle through a multi-modality, synergistic combination of pressurized topical oxygen and cyclical compression, delivered conveniently at home.
Today, the evidence supporting TWO₂ is stronger than ever.
Why Combining Randomized Controlled Trial Data and Real-world Evidence Matters
RCTs provide gold-standard proof of safety and efficacy through controlled, bias-reduced study design but they often include only a narrow slice of patients.
RWE shows how treatments perform in everyday clinical practice across broader, more diverse populations, real-world behaviors, and long-term use.
By combining both, you get evidence that’s strong, representative, and reflective of how patients respond in real life, supporting better clinical decisions and better outcomes.
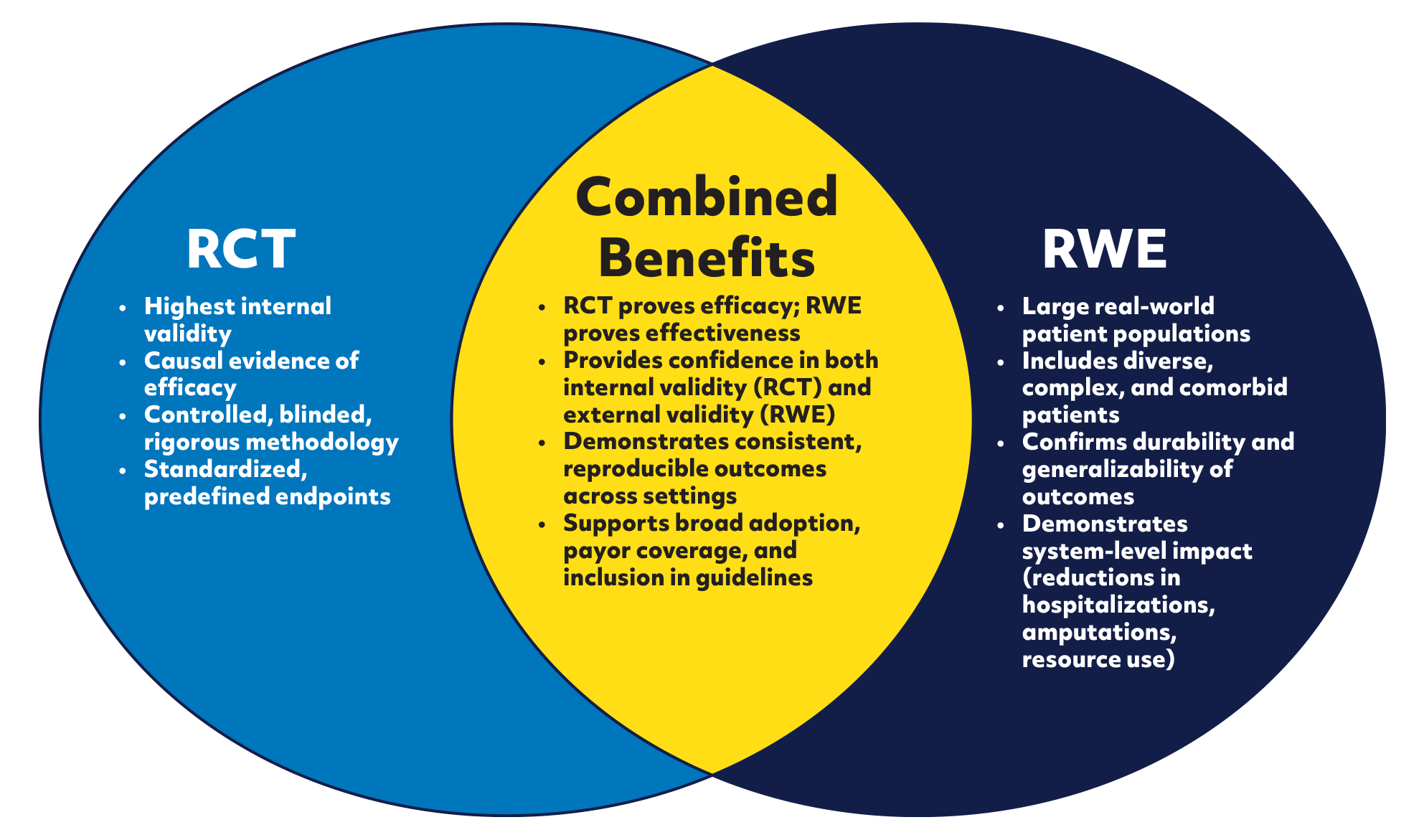
Our proof: RCT Rigor + Real-World Scale
Randomized Controlled Evidence (RCT) – Demonstrating Causation
The landmark TWO₂ RCT1 demonstrates that TWO₂ therapy delivers significantly higher healing rates, faster closure, and dramatically lower recurrence compared with standard care or sham controls. In diabetic foot ulcers, TWO₂ achieved:
- 2–3x higher healing rates than control arms (41.7% closure rate at 12 weeks vs 13.5% in the control group (P = .004))
- Sustained benefit at 12 months, with recurrence rates as low as 6–7% versus ~40% in controls.
- Similarly, Armstrong et al2, published 40% of all patients with DFUs experience a recurrence within 1 year after the ulcer has healed as seen in the RCT controls.
- Consistently awarded with the lowest risk of bias score, utilizing the respected Cochrane Collaboration’s tool for assessing risk of bias in RCTs
Real-World Evidence (RWE) – Demonstrating Generalizability and Impact at Scale
A well-designed retrospective, matched cohort study of TWO2 in the treatment of DFUs in veterans by Yellin, et al.3, demonstrated:
- 82% reduction in hospitalizations (7.1% vs 40%; P < .0001) at 12 months
- 73% reduction in amputations (8.6% vs 31.4%; P = .0007) at 12 months
The recurrence rate for VLU is reported to be between 24%-57% at 12 months.4,5 Tawfick, et al6, in a prospective controlled study, comparing ITOT with conventional compression dressings (CCD) in the management of nonhealing VLUs, present for greater than two years demonstrated:
- Wound recurrence rate of only 6% (3 of 51) vs. a 47% (14 of 30) recurrence rate in the CCD group at 36 months
A large cohort (3126-patient), multicenter real-world study further validated TWO₂ healing in complex, comorbid populations. Even in patients who had failed Standard of Care and other advanced therapies, outcomes exceeded those seen in national wound registries, even in less complex patient groups.
In this study healing rates were:
- 64.8% overall healing in a mean time of 4.2 months, despite wounds being chronic for an average of 7 months before starting therapy
- 63% healing in DFUs in a mean time of 18 weeks
- 72% in VLUs in a mean time of 17 weeks
- Consistent benefit across DFU, VLU, arterial, and atypical wounds
Cost Effectiveness Analysis
A formal cost-effectiveness analysis of ITOT for DFUs using National Health Service costing norms in England showed that TWO2 both increased quality-adjusted life years and reduced overall treatment costs.7
Together, the evidence tells a clear story
Across rigorous trials, large-scale real-world cohorts, and analysis studies, TWO₂ therapy consistently delivers:
- Superior clinical outcomes
- Durable tissue quality and dramatically reduced recurrence
- Lower amputation and hospitalization rates
- High tolerability and patient adherence
- Demonstrated cost savings in health-economic evaluations
TWO2 therapy demonstrates both proven efficacy and real-world effectiveness, consistently delivering superior healing, reduced complications, improved patient quality of life, and health-economic value. For payors, providers, and regulators alike, the evidence supports TWO2 as a safe, effective, home-based, and economical adjunctive therapy that merits broad adoption, reimbursement, and integration into standard-of-care pathways for chronic wounds.
Outcomes
TWO2 therapy delivers demonstrated real-world reductions in hospitalizations and amputations, even in complex, comorbid populations—outcomes that translate into cost savings, improved patient quality of life, and greater limb-salvage. For payors, providers, and regulators alike, the evidence supports TWO2 as a safe, effective, home-based, and economical adjunctive therapy that merits broad adoption, reimbursement, and integration into standard-of-care pathways for diabetic foot ulcers.
Methodological rigor/data integrity
TWO₂ therapy is supported by a rigorous evidence foundation in wound care, combining independent meta-analytic validation with a multinational, multicenter, randomized, double-blind, placebo-controlled trial, the highest standard of clinical research.
“Chronic wounds persist due to a self-sustaining cycle of hypoxia, edema, and inflammation. By integrating oxygen delivery with cyclical compression, ITOT [TWO2] directly addresses the multifactorial barriers to repair, promoting durable healing and reducing complications. This multi-modality approach represents a promising therapeutic advance in the management of refractory lower extremity wounds, with broad implications for improving outcomes and quality of life and reducing health care costs.8”
Reference:
- Frykberg RG, Franks PJ, Edmonds M, et al., A Multinational, Multicenter, Randomized, Double-Blinded, Placebo-Controlled Trial to Evaluate the Efficacy of Cyclical Topical Wound Oxygen (TWO2) Therapy in the Treatment of Chronic Diabetic Foot Ulcers: The TWO2 Study. Diabetes Care. 2020;43(3):616-624. doi:10.2337/DC19-0476
- Armstrong DG, Boulton AJM, Bus SA. Diabetic Foot Ulcers and Their Recurrence. Ingelfinger JR, editor. New England Journal of Medicine. 2017 Jun;376(24):2367–75. http://www.nejm.org/doi/10.1056/NEJMra1615439)
- Yellin JI, Gaebler JA, Zhou FF, et al., Reduced Hospitalizations and Amputations in Patients with Diabetic Foot Ulcers Treated with Cyclical Pressurized Topical Wound Oxygen Therapy: Real-World Outcomes. Adv Wound Care (New Rochelle). 2022;11(12):657-665. doi:10.1089/wound.2021.0118
- McDaniel HB, Marston WA, Farber MA, Mendes RR, Owens L V., Young ML, et al., Recurrence of chronic venous ulcers on the basis of clinical, etiologic, anatomic, and pathophysiologic criteria and air plethysmography. J Vasc Surg. 2002;35(4):723–8. https://pubmed.ncbi.nlm.nih.gov/11932670/
- Finlayson K, Wu ML, Edwards HE. Identifying risk factors and protective factors for venous leg ulcer recurrence using a theoretical approach: A longitudinal study. Int J Nurs Stud [Internet]. 2015 Jun 1;52(6):1042–51. https://pubmed.ncbi.nlm.nih.gov/25801312/
- Tawfick WA, Sultan S. Technical and Clinical Outcome of Topical Wound Oxygen in Comparison to Conventional Compression Dressings in the Management of Refractory Nonhealing Venous Ulcers. Vasc Endovascular Surg. 2013;47(1):30-37. doi:10.1177/1538574412467684
- Kerr, M., Wild, D., Edmonds, M., et al., Cost effectiveness of topical wound oxygen therapy for chronic diabetic foot ulcers J Diabetes Complications. 2025; 39, 109016
- Lohr JM, Raffetto JD, Dexter DJ, Regulski MJ, Edmonds ME, Ozsvath KJ, Blakely MM. A synergistic multimodality treatment approach to address the key drivers of wound chronicity. J Vasc Surg Venous Lymphat Disord. 2025 Oct 23;14(1):102348. doi: 10.1016/j.jvsv.2025.102348. Epub ahead of print. PMID: 41138912; PMCID: PMC12670913